 Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.  Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.
 Offering SPR-BLI Services - Proteins provided for free!
Offering SPR-BLI Services - Proteins provided for free! Get your ComboX free sample to test now!
Get your ComboX free sample to test now!
 Time Limited Offer: Welcome Gift for New Customers !
Time Limited Offer: Welcome Gift for New Customers !  Shipping Price Reduction for EU Regions
Shipping Price Reduction for EU Regions
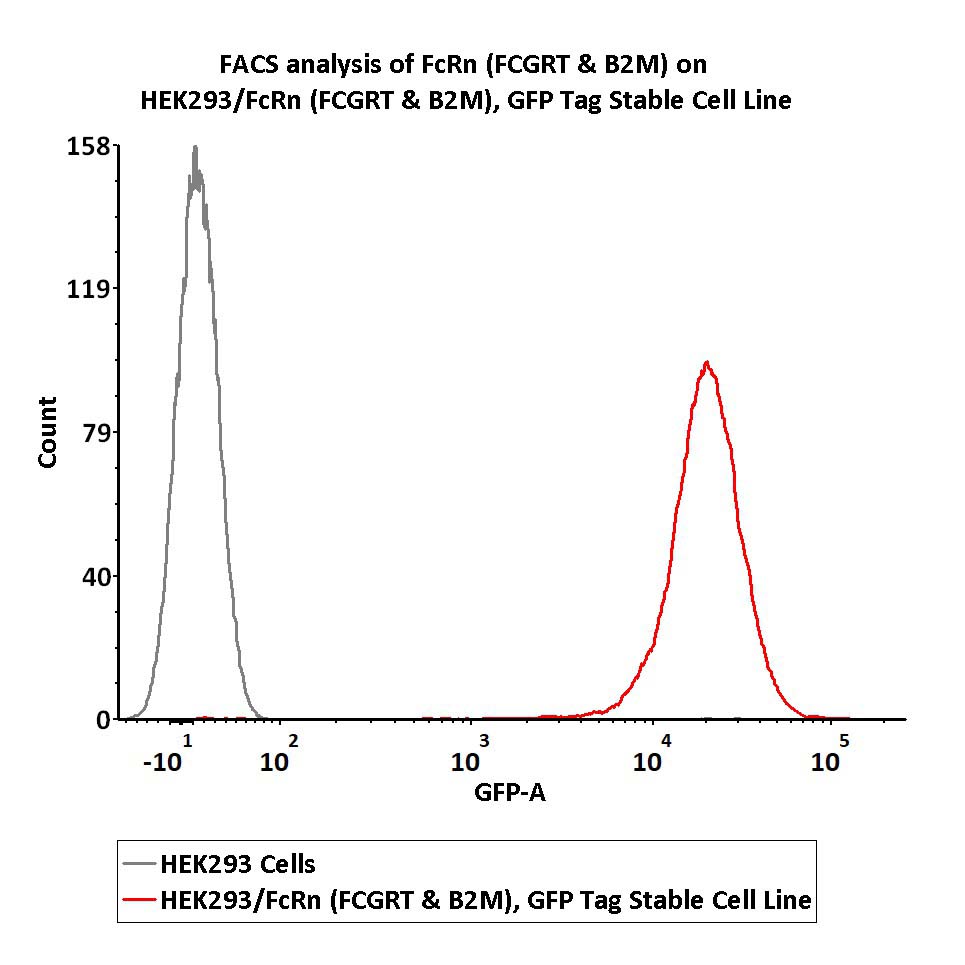
Fig. FACS analysis of FcRn (FCGRT & B2M) on HEK293/FcRn (FCGRT & B2M), GFP Tag Stable Cell Line.
HEK293/FcRn (FCGRT & B2M), GFP Tag Stable Cell Line was red line, negative control HEK293 cells was grey line(QC tested).
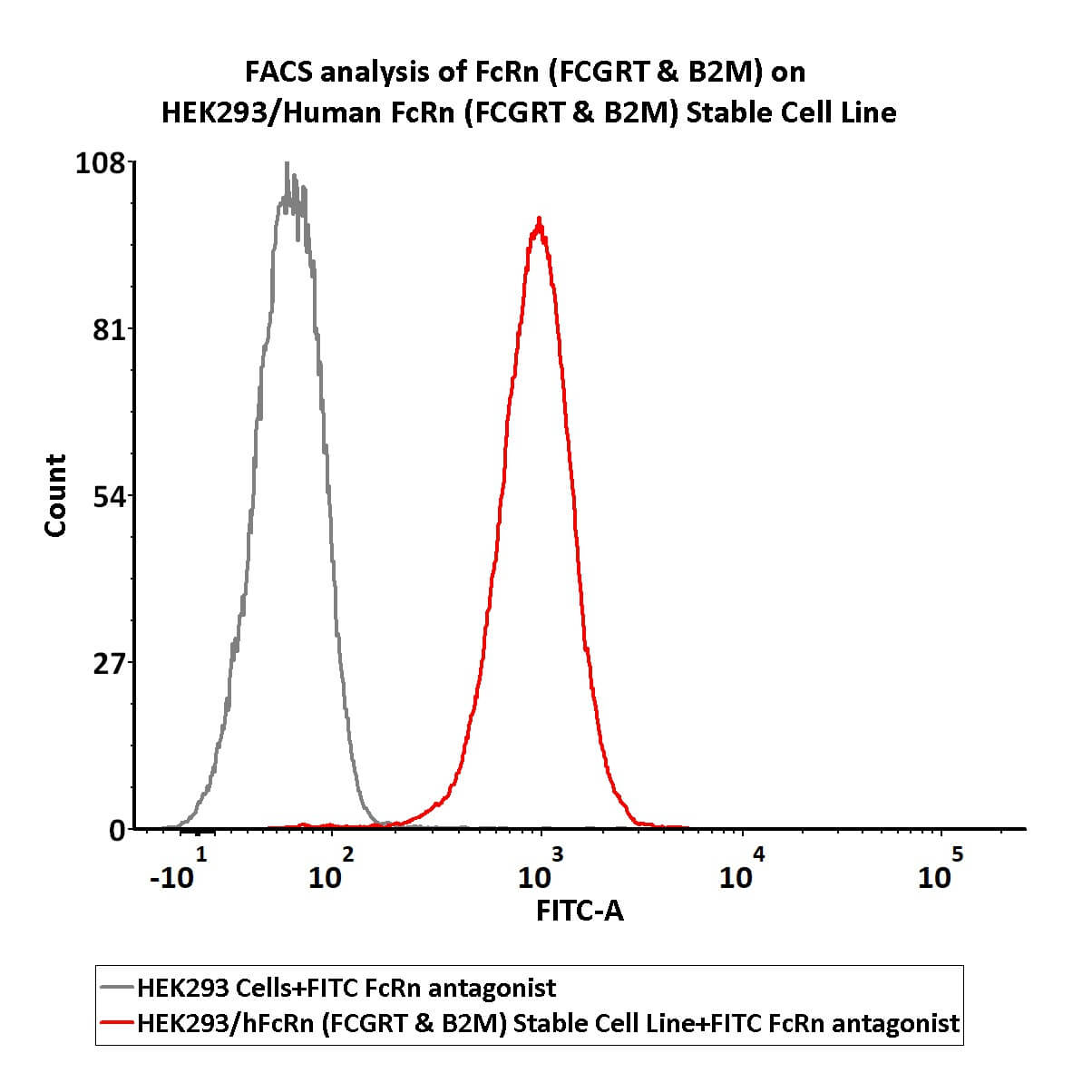
FACS analysis of FcRn (FCGRT & B2M) on HEK293/Human FcRn (FCGRT & B2M) Stable Cell Line.
FACS assay shows that FITC-Labeled Human IgG1 Fc (C103S, M135Y, S137T, T139E, H316K, N317F) Protein, His Tag (Cat. No. IG1-HF2H3) can bind to HEK293/Human FcRn (FCGRT & B2M) Stable Cell Line. HEK293/Human FcRn (FCGRT & B2M) Stable Cell Line was red line, negative control HEK293 cells was grey line(QC tested).
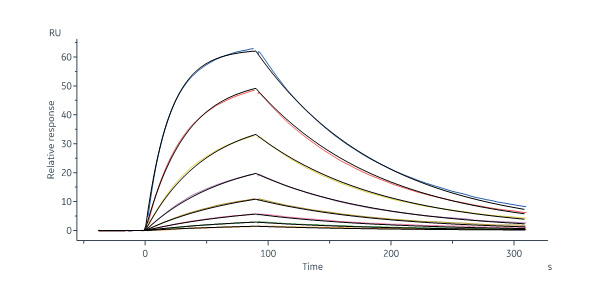
Human IgG1 Fc (C103S, M135Y, S137T, T139E, H316K, N317F), His Tag (Cat. No. IG1-H52H8) immobilized on CM4 Chip can bind Human FCGRT&B2M Heterodimer Protein, Strep Tag&Tag Free (Cat. No. FCM-H5283) with an affinity constant of 23.8 nM as determined in a SPR assay (Biacore 8K) (QC tested).
| Name | Research Code | Research Phase | Company | First Brand Name | First Approved Country | First Indication | First Approved Company | First Approved Date | Indications | Clinical Trials |
|---|---|---|---|---|---|---|---|---|---|---|
| Efgartigimod alfa | ARGX-113; EFG PH20; EFG PH20 SC; PH20 SC | Approved | Argenx Se | VYVGART HYTRULO, Vyvgart, 卫伟迦, VYVGART | United States | Myasthenia Gravis | Argenx BV | 2021-12-17 | Postural Orthostatic Tachycardia Syndrome; Tachycardia; Pemphigus; Nephrotic Syndrome; Muscular Diseases; Guillain-Barre Syndrome; Thrombocytopenia; Pemphigoid, Bullous; Glomerulonephritis, Membranous; Graves Ophthalmopathy; Lupus Nephritis; Polyradiculoneuropathy, Chronic Inflammatory Demyelinating; Sjogren's Syndrome; Autoimmune Diseases; Polyneuropathies; Nephrosis; Dermatomyositis; Myositis; Polymyositis; Myasthenia Gravis; Purpura, Thrombocytopenic, Idiopathic | Details |
| Rozanolixizumab | UCB-7665 | Approved | Ucb Sa | RYSTIGGO | United States | Myasthenia Gravis | Ucb Inc | 2023-06-27 | Polyradiculoneuropathy, Chronic Inflammatory Demyelinating; Purpura, Thrombocytopenic, Idiopathic; Myasthenia Gravis; Thrombocytopenia; Glioma; Fibromyalgia; Encephalitis | Details |
| Efgartigimod alfa/Hyaluronidase | Approved | Argenx BV | VYVDURA, VYVGART HYTRULO | United States | Myasthenia Gravis | Argenx BV | 2023-06-20 | Myasthenia Gravis | Details | |
| Efgartigimod alfa | ARGX-113; EFG PH20; EFG PH20 SC; PH20 SC | Approved | Argenx Se | VYVGART HYTRULO, Vyvgart, 卫伟迦, VYVGART | United States | Myasthenia Gravis | Argenx BV | 2021-12-17 | Postural Orthostatic Tachycardia Syndrome; Tachycardia; Pemphigus; Nephrotic Syndrome; Muscular Diseases; Guillain-Barre Syndrome; Thrombocytopenia; Pemphigoid, Bullous; Glomerulonephritis, Membranous; Graves Ophthalmopathy; Lupus Nephritis; Polyradiculoneuropathy, Chronic Inflammatory Demyelinating; Sjogren's Syndrome; Autoimmune Diseases; Polyneuropathies; Nephrosis; Dermatomyositis; Myositis; Polymyositis; Myasthenia Gravis; Purpura, Thrombocytopenic, Idiopathic | Details |
| Rozanolixizumab | UCB-7665 | Approved | Ucb Sa | RYSTIGGO | United States | Myasthenia Gravis | Ucb Inc | 2023-06-27 | Polyradiculoneuropathy, Chronic Inflammatory Demyelinating; Purpura, Thrombocytopenic, Idiopathic; Myasthenia Gravis; Thrombocytopenia; Glioma; Fibromyalgia; Encephalitis | Details |
| Efgartigimod alfa/Hyaluronidase | Approved | Argenx BV | VYVDURA, VYVGART HYTRULO | United States | Myasthenia Gravis | Argenx BV | 2023-06-20 | Myasthenia Gravis | Details |
| Name | Research Code | Research Phase | Company | Indications | Clinical Trials |
|---|---|---|---|---|---|
| Nipocalimab | M-281; JNJ-80202135 | Phase 3 Clinical | Momenta | Polyradiculoneuropathy, Chronic Inflammatory Demyelinating; Myasthenia Gravis; Hemolysis; Myositis; Arthritis, Rheumatoid; Sjogren's Syndrome; Lupus Nephritis; Anemia, Hemolytic, Autoimmune; Lupus Erythematosus, Systemic; Pemphigoid, Bullous | Details |
| IMVT-1402 | HL-161 ANS; IMVT-1402 | Phase 1 Clinical | Immunovant Sciences Gmbh, HanAll Biopharma Co Ltd | Autoimmune Diseases | Details |
| Nipocalimab | M-281; JNJ-80202135 | Phase 3 Clinical | Momenta | Polyradiculoneuropathy, Chronic Inflammatory Demyelinating; Myasthenia Gravis; Hemolysis; Myositis; Arthritis, Rheumatoid; Sjogren's Syndrome; Lupus Nephritis; Anemia, Hemolytic, Autoimmune; Lupus Erythematosus, Systemic; Pemphigoid, Bullous | Details |
| IMVT-1402 | HL-161 ANS; IMVT-1402 | Phase 1 Clinical | Immunovant Sciences Gmbh, HanAll Biopharma Co Ltd | Autoimmune Diseases | Details |
This web search service is supported by Google Inc.
